Feds issue new guidelines, launch probe to address early allergic reactions to COVID vaccine
CDC guidelines say anyone who has suffered severe reaction should skip second dose. Chemical known as polyethylene glycol suspected.
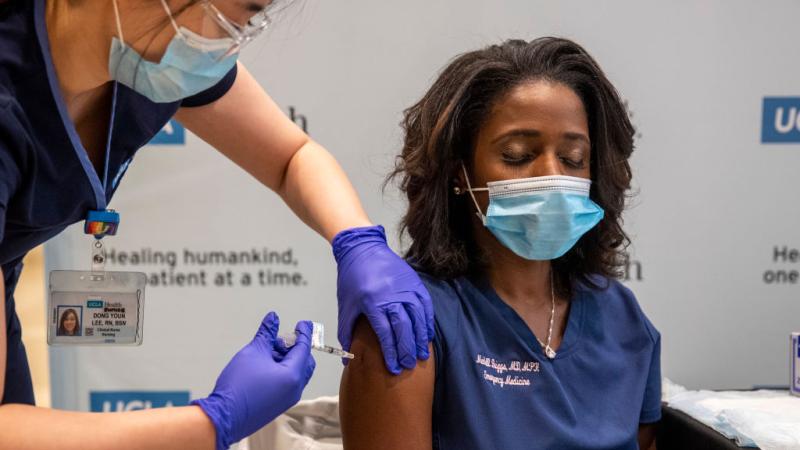
The Centers for Disease Control and Prevention (CDC) issued new guidelines and the Food and Drug Administration launched an investigation after a half dozen early patients suffered severe allergic reactions after getting the COVID-19 vaccine.
The CDC said those who suffered a severe allergic reaction known as anaphylaxis – which required hospitalization or an EpiPen treatment --after the first dose of the vaccine should not get the second shot.
And it said people who have had severe allergic reactions to a component in a COVID-19 vaccine should not get that specific vaccine. There are currently two approved vaccines, one from Pfizer and the other from Moderna.
“CDC recommends that people with a history of severe allergic reactions not related to vaccines or injectable medications—such as allergies to food, pet, venom, environmental, or latex—may still get vaccinated," the CDC said. "People with a history of allergies to oral medications or a family history of severe allergic reactions, or who might have a milder allergy to vaccines (no anaphylaxis)—may also still get vaccinated.”
You can read the full CDC guidelines here.
Meanwhile, Peter Marks, the director of the FDA’s Center for Biologics Evaluation and Research, said the agency was investigating about a half dozen severe reactions to Pfizer’s vaccine to determine whether a chemical known as polyethylene glycol “could be the culprit.”
The chemical is in both approved vaccines.
You can listen to more of Marks’ presentation here.