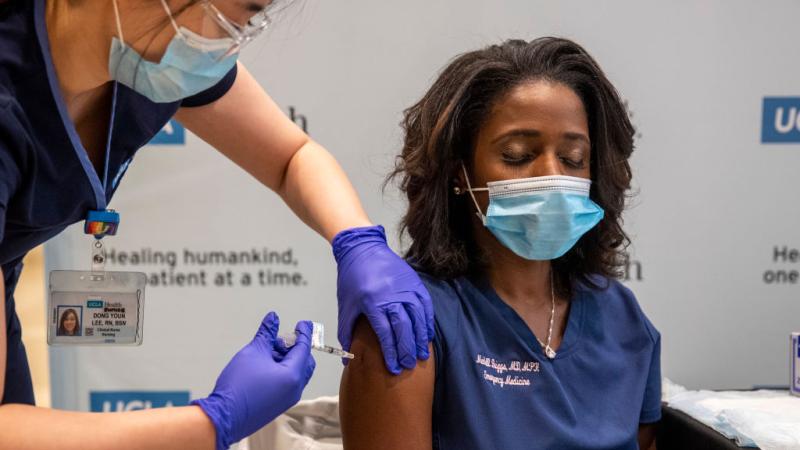
Moderna applies for full FDA approval of its COVID-19 vaccine
If approved the vaccine will stay on U.S. markets after the pandemic is over.
Moderna on Monday asked the Food and Drug Administration for full approval of its COVID-19 vaccine.
The move makes Moderna the second U.S. drugmaker to seek a biologics license that will allow it to market the shots directly to customers, according to CNBC.
Full approval will allow the vaccine to stay on the U.S. market once the pandemic is over.